maxresorb®
Innovative biphasic calcium phosphate

Very rough and hydrophilic surface
100% synthetic and resorbable
IDEAL OSTEOCONDUCTIVE PROPERTIES
maxresorb® is an innovative, safe, reliable, and fully synthetic bone substitute material that is characterized by controlled resorption and outstanding handling characteristics. maxresorb® is composed of 60% slowly resorbing hydroxyapatite (HA) and 40% fast resorbing beta-tricalcium phosphate (β-TCP) 1,2. The unique synthesis-based production process ensures a completely homogenous distribution of both mineral phases. The special composition of maxresorb® promotes fast formation of new vital bone, and ensures a controlled resorption without volume loss of the augmented site.
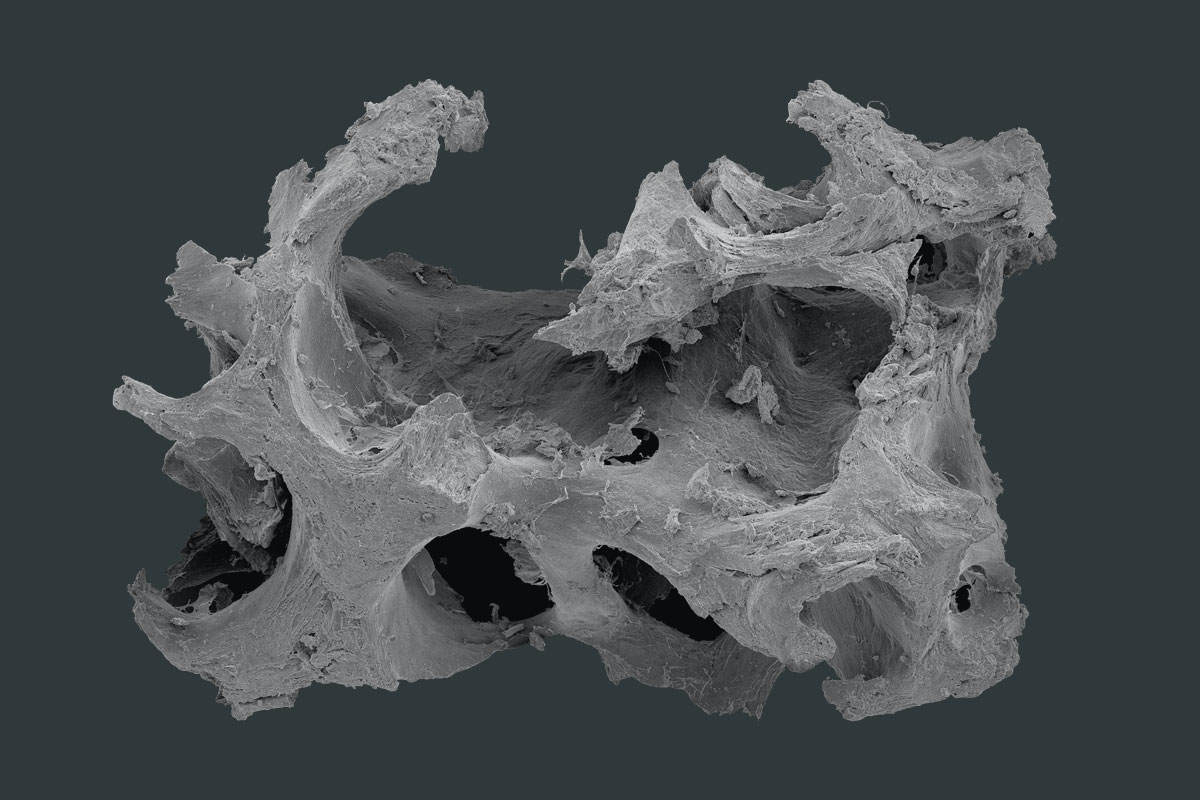
ULTRA-HIGH INTERCONNECTED POROSITY
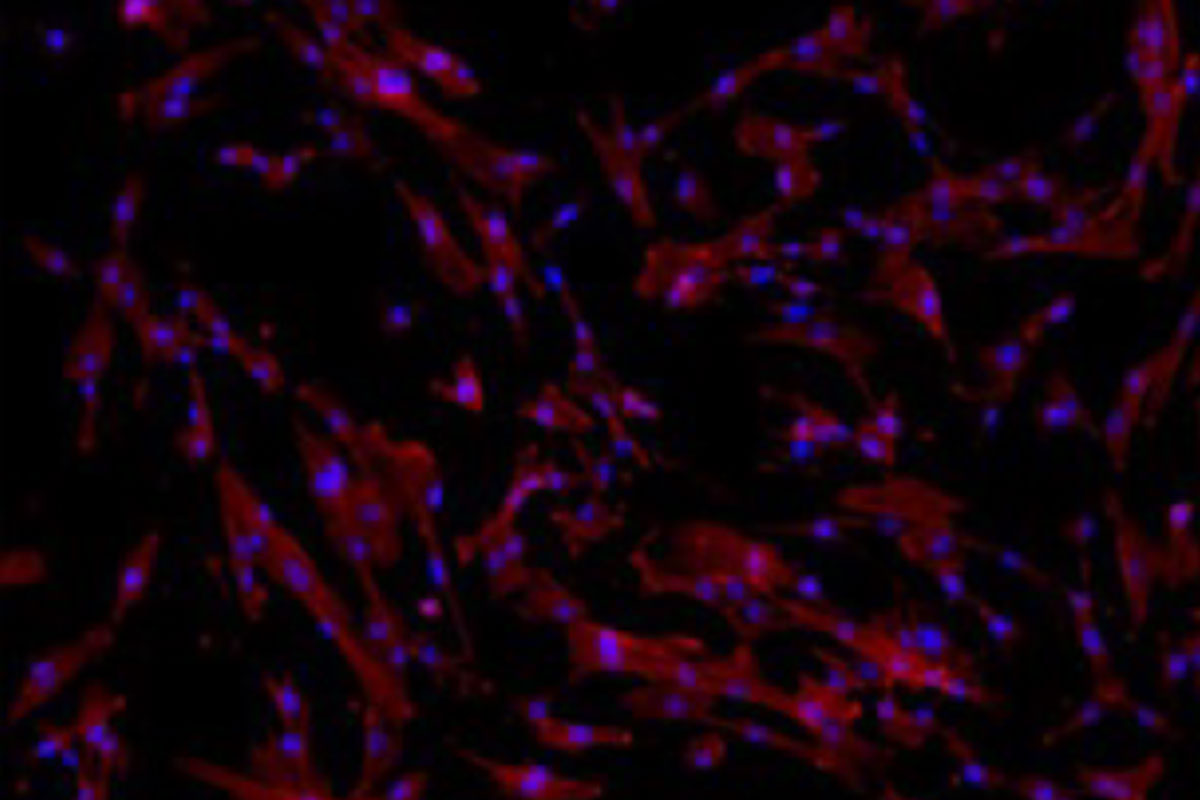
The osteoconductivity of maxresorb® is based on a network of interconnecting pores, a very high overall porosity of approx. 80% as well as its very rough surface 3, 4, 5. The nano-structured surface facilitates the adsorption of blood, proteins, and stem cells and promotes cell differentiation and osseous integration. maxresorb® is thus an ideal scaffold for the migration of bone forming cells and binding of signaling molecules, which can accelerate tissue integration and regeneration 6, 7.
maxresorb® offers a reliable alternative to xenogeneic bone substitute materials in a variety of indications.
IMPLANTOLOGY, PERIODONTOLOGY AND ORAL AND CMF SURGERY
- Sinus lift
- Ridge augmentation
- Intraosseous defects
- Extraction sockets
- Osseous defects
- Furcation defects
Choice of particle size
Small maxresorb® particles (0.5 – 1.0 mm) allow good adaptation to surface contours; they are especially useful for lateral augmentations or to fill voids when working with autologous bone blocks. For sinus lift and extensive augmentations the use of large maxresorb® particles (0.8 – 1.5 mm) is recommended. The increased space between the large particles enables a better vascularization and improves the regeneration of larger defect
Hydration
maxresorb® granules can be applied dry or wet. Hydration in blood or sterile saline solution facilitates the handling and application of the granules, as they adhere to each other. The excellent hydrophilicity of maxresorb® permits rapid hydration.
Compaction of the granules
To provide space for the regenerative process, only gentle compression of the particles is recommended. It allows for revascularization and osseointegration of the bone grafting particles by the three-dimensional pore network of maxresorb®.
Stabilization of the granules
maxresorb® granules should always be covered by a membrane in order to immobilize the particles at the augmented site and to prevent soft tissue ingrowth into the bone defect. When treating defects outside the ridge contour, a fixation of the membrane by pins or screws can be advantageous.
Healing time and re-entry
Although clinical results demonstrate a somewhat faster regeneration of defects treated with maxresorb®, it is recommended to follow the same treatment protocol as for bovine materials. Accordingly, re-entry after ridge augmentation or sinus lift procedures with maxresorb® should be performed 6 months post-operatively. This time can be shortened for the regeneration of smaller defects or by mixing with autologous/allogenic bone.
Mixing with autologous bone
Mixing of maxresorb® with autologous bone adds a biological activity (osteoinductive and osteogenetic properties of autologous bone) and support faster regeneration and improved formation of new bone.
- 60% HA/40% β-TCP
- Osteoconductive
- Ultra-high interconnected porosity
- Volume and mechanical graft stability
- Very rough and hydrophilic surface
- 100% synthetic and resorbable
- maxresorb® product brochure
- FAQ maxresorb®
- maxresorb® Literture list
- EC-Certificate EC Design Examination Certificate 93/42/EEC maxresorb®
- Patient information bone augmentation with biomaterials
- Patient information socket preservation
- botiss product portfolio
- Summary of safety and clinical performance
Distribution
With our international network of distribution partners, we are near you in over 100 countries worldwide. In addition to our 360° productportfolio, we offer service, scientific advice and exchange, training and events directly on site from a single source.
Find a distribution partner near you: